HumanKine® Recombinant proteins
Cytokines and growth factors are essential to advanced therapies. Unlike traditional pharmacologics, cell and gene therapies require the expansion and maintenance of living cells. These recombinant proteins coordinate and sustain these processes.
“FOR HUMANS...BY HUMAN CELLS”
With the rise in clinical trials, there is a greater demand for safe, pure, and potent recombinant proteins. Xeno-free and animal component-free, Proteintech’s HumanKine recombinant proteins are ISO 13485 certified and GMP-compliant, making them ideal for clinical applications.
Proteintech Group manufactures RUO and GMP-grade HumanKine recombinant proteins using a human expression system.
Recombinant proteins created in HEK293 cells using animal-free components offers the following advantages when compared with proteins manufactured using bacterial based platforms:
- High bioactivity & stability
- Lot-to-lot consistency
- Native human conformation & post-translational modifications
- Endotoxin-free, xeno-free & tag-free

A comprehensive overview of the benefits of HumanKine HEK293-expressed human recombinant proteins:
1. Animal component, endotoxin, and xeno free
Products derived from or by an animal are not used at any point during production. The benefits of animal component free and xeno free proteins are as following:
- The final product does not contain any constituent or components from non-human derivation.
- All the materials from procurement to final products are stored and handled in dedicated animal free facility.
- Final product or the process does not involve the use of materials or recombinant materials from non-human sources.
- Besides using a human expression system to minimize endotoxin levels, each lot is verified to be endotoxin free by LAL assay
2. Tag-free
HumanKine recombinant proteins are expressed and purified without any tags. There are several benefits of tag free proteins:
- Inclusion of a tag can often result in changes to the structure of the protein of interest.
- A tag may interfere with the active site of the protein resulting in altered biological activity.
- Presence of a tag can increase immunogenicity of some proteins, which makes a tag- free recombinant protein more desirable and reliable for in vivo applications.
3. Native folding and maturation
Proper folding and maturation are crucial for optimal protein activity. Chinese Hamster Ovary (CHO) cells are widely used as a mammalian expression system for large-scale protein production, however they lack certain human-specific chaperones. In contrast, HumanKine recombinant proteins are expressed in a human cell line, HEK293. This system has the optimal machinery for optimizing conformation and stability of human recombinant proteins. For example, Figure 1 demonstrates how this system generates more mature Activin A in comparison to the conventional CHO cell expression system.

4. Authentic human glycosylation
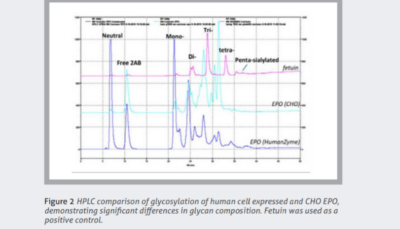
HumanKine recombinant proteins are produced only in HEK293 cells. As a human cell line, this system generates glycoproteins that more closely resemble naturally occurring proteins, optimizing safety and potency. Figure 2 highlights the differences in glycan profiles of EPO recombinant protein expressed in CHO cells and HumanKine human expression systems.
5. High Activity
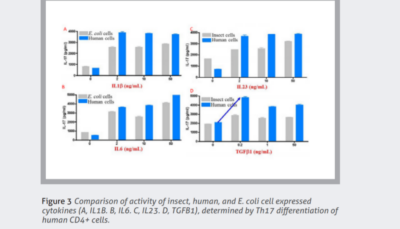
Tag-free proteins expressed in HEK293 cells create proteins that tend to have higher activity than those produced in other expression systems. Figure 3 compares activity for multiple cytokines between different eukaryotic systems.
Scale up from pre-clinical to clinical with Proteintech:
HumanKine RUO and GMP cytokines and recombinant proteins are both manufactured using the same expression systems, production process and operating procedures in a GMP facility. This ensures there is no variability in the quality and performance of these products, facilitating the smooth transition from pre-clinical to clinical
Please contact Jody Salomon for more information about Cytokines and Recombinant Proteins.
Whitepaper library
Several white papers are published covering HumanKine cytokines and recombinant proteins:
- Time-Saving Dendritic Cell Generation No Medium Change - Less Reagents With HumanKine® GM-CSF and HumanKine IL4
- HumanKine® IL-23 100x More Potent For Induction of Human Th17 Cells
- Effective Polarization of Human Th17 Cells with Biologically Relevant HumanKine® TGFβ1, TGFβ2, and TGFβ3 Expressed in Human Cells
- Efficient Induction of Th17 Cells with HumanKine® TGFβ1
- Native Glycosylation Is Important For Biological Function and Activity of Recombinant Human Proteins
- HumanKine® IL-17 Effective Tool for Autoimmune Disease Research
- Effective Differentiation of Th17 Cells With HumanKine® Cytokines Expressed in Human Cells
- HumanKine® Noggin Human Cell-expressed Glycosylated Disulfide-linked Homodimer
