Sanbio, exclusive distributor of the Benelux!
Cayman Chemical Forensic
Forensic Science Products.
Sanbio BV is in possession of an opium exemption for trade-related purposes with register number 16455 07 HD.
About us
Cayman Chemical Forensic
With more than 30 years of experience in the synthesis, purification, and characterization of biochemicals, Cayman Chemical has become a leader in the field of designer drugs by providing high-purity reference standards to federally licensed laboratories and qualified academic research institutions for forensic analysis. Cayman’s highly trained staff of chemists provide institutions with solutions to quickly identify and understand the physiological and toxicological properties of new designer drugs. Cayman synthesizes a range of analytical standards including synthetic cannabinoids, cathinones, phenethylamins, amphetamines, indanes, opioids, benzodiazepines, tryptamines, and phytocannabinoids, among many others.
“We are committed to ensuring and maintaining quality in all aspects of our reference material production..,,
Reference Material (RM)
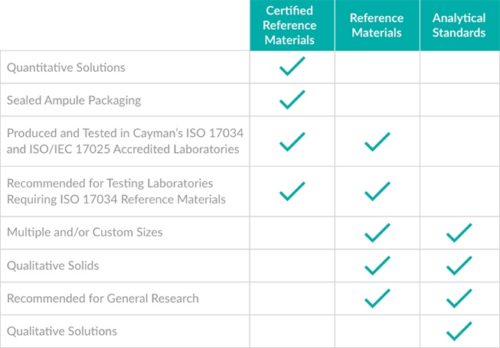
Cayman’s Reference Materials (RMs) are sold in several sizes allowing for a low-cost, scalable, and highly flexible option for analytical labs. An RM is fully characterized, and its property values are tested under an ISO/IEC 17025:2017 accredited quality system, making it fit for direct use by labs requiring traceable standards.
Certified Reference Material (CRM)
Certified Reference Materials (CRMs) serve as Primary Standards as defined by ISO and are suitable for labs requiring traceable quantitative standards. A CRM, as defined by ISO 17034:2016, must report the certified concentration and corrected purity values with the corresponding measurement uncertainties. Cayman’s CRMs ensure the metrological traceability and accuracy needed to perform quantitative measurements and are sold as quantitative solutions in sealed ampules.
Analytical Standards
Cayman’s Forensic Analytical Standards are accompanied by testing documentation to demonstrate purity and identity characterization and are formulated in small quantities as neat solids or in solution for use in analytical testing. They may be suitable for application in identification, analytical method validation, and quantification. However, analytical methods requiring quantification may require assessment of suitability or in-house verification to meet laboratory specifications and accreditations. Most Reference Materials are initially produced as Analytical Standards in order to make the Reference Material quickly available for use.
Quality
Which quality grade is right for you?
Quality and Certifications
For institutions that rely on highly traceable reference materials for use in quantitative or qualitative testing, Cayman offers synthesized reference materials and certified reference materials accredited to meet ISO/IEC 17025:2017 and ISO 17034:2016 standards.
Documentation
Each RM and CRM is accompanied by enhanced documentation conforming to ISO standards for testing laboratories and RM producers. Analytical Standards are accompanied by Cayman’s standardized product information.
Cayman’s RMs will include a Confirmation of Analysis (CofA) document that displays a comprehensive table of methods used for the analysis of the neat material, an estimate of uncertainty for each measurement, all relevant analytical data, and a stability assessment.
Cayman’s CRMs will include a Certificate of Analysis (CofA) that reports the certified concentration and corrected purity values with the corresponding measurement uncertainties in addition to displaying a comprehensive table of methods used for the analysis of the neat material, an estimate of uncertainty for each measurement, all relevant analytical data, and a stability assessment.
Cayman’s Analytical Standards will include a Certificate of Analysis (CofA) that reports identity, purity, and other relevant characteristics, displaying a minimum of two purity and two identity characterization tests.
Please contact our support department for more information about Forensic Science products.