-
Research area
- Biochemicals
- Blood and Biospecimens
- Cell biology
- Environmental
- Flow Cytometry
- Forensic Science
- Genomics
- Immunology
- Labware
- Microbiology
- Pathology
- Transplantation
429 Too Many Requests 429 Too Many Requests
nginx - Suppliers
- About us
- Resources
- Events
- Support
- Lab Services
Product description
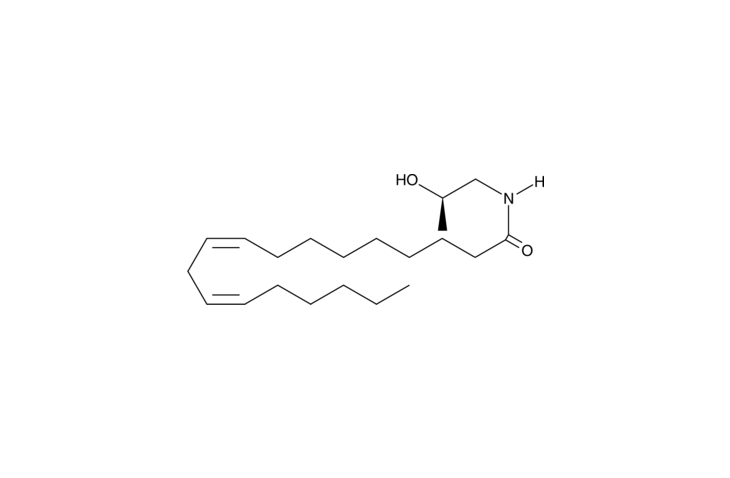
N-Acyl ethanolamines (NAEs) have diverse biological actions that are strongly affected by the associated acyl group. Linoleoyl ethanolamide (LOEA) has potential signaling roles in aging and neurological functioning.{19623,11177} LOEA has a weak affinity for cannabinoid (CB) receptors (Ki = 10, 25 ?M for CB1, CB2, respectively) and inhibits voltage-gated K+ channels.{7422,13859} LOEA also inhibits fatty acid amide hydrolase (FAAH; Ki = 9 ?M) and is hydrolyzed by FAAH.{7251,3922} (R)-(-)-Linoleyl-2’-hydroxy-1’-propylamide is a homolog of LOEA which is characterized by the addition of an (R)-B-methyl group at the terminal ethanolamine carbon. A similar modification of arachidonoyl ethanolamide (Item No. 90050) to produce R-2 methanandamide (Item No. 90074) imparts diminished affinity for the CB receptor as well as reduced metabolic stability.{1092} The physiological actions of this compound have not been evaluated.
Specifications
Supplier
Cayman Chemical
Shipping & storage
Shipping condition
Dry Ice
Storage temperature
-20°C
Do you have any questions about this product?
Order your product by email
Productname
(R)-(-)-Linoleyl-2'-Hydroxy-1'-Propylamide
9001234-1
By filling out this form, you are placing an order by e-mail. You will receive an order confirmation within one working day. The order cannot be modified after receipt of the order confirmation.
Request a sample
Productname
(R)-(-)-Linoleyl-2'-Hydroxy-1'-Propylamide
9001234-1
By filling out this form, you request a sample. You will receive an order confirmation within one working day. The order cannot be modified after receipt of the order confirmation.
Are you looking for specific products, alternatives or documentation?