Product description
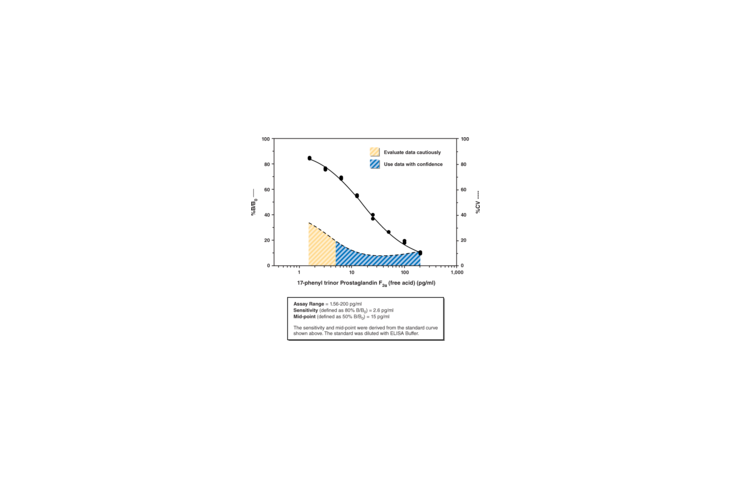
17-phenyl trinor Prostaglandin F2a (17-phenyl trinor PGF2a) is a metabolically stable analog of PGF2a and is a potent agonist for the FP receptor. It binds to the FP receptor on ovine luteal cells with a relative potency of 756% compared to that of PGF2a.{2058} At the rat recombinant FP receptor expressed in CHO cells, 17-phenyl trinor PGF2a inhibits PGF2a binding with a Ki value of 1.1 nM.{1374} In human and animal models of glaucoma, FP receptor agonist activity corresponds very closely with intraocular hypotensive activity. 17-phenyl trinor PGF2a ethyl amide is an F-series prostaglandin analog which has been approved for use as an ocular hypotensive drug, sold under the Allergan trade name Bimatoprost.{8941} Whether 17-phenyl trinor PGF2a ethyl amide is a prodrug analogous to prostaglandin ester prodrugs such as latanoprost is currently controversial. There is some evidence that unmetabolized 17-phenyl trinor PGF2a is a weak FP receptor agonist.{9581} 17-phenyl trinor PGF2a is converted by an amidase enzymatic activity in the bovine and human cornea to yield the corresponding free acid, with a conversion rate of about 40 µg/g corneal tissue/24 hours.{9311} Cayman’s 17-phenyl trinor PGF2a ELISA is a sensitive detection method for measuring both the free acid and ethyl amide forms of 17-phenyl trinor PGF2a. The assay is most appropriate for use when only one of the two forms is present. Samples containing mixtures of both the ethyl amide and free acid should be purified and the two compounds separated prior to ELISA analysis.
Specifications
Supplier
Cayman Chemical
Shipping & storage
Shipping condition
Dry Ice
Storage temperature
-20°C
Do you have any questions about this product?
Order your product by email
Productname
17-phenyl trinor Prostaglandin F2a ELISA Kit
516821-480
By filling out this form, you are placing an order by e-mail. You will receive an order confirmation within one working day. The order cannot be modified after receipt of the order confirmation.
Request a sample
Productname
17-phenyl trinor Prostaglandin F2a ELISA Kit
516821-480
By filling out this form, you request a sample. You will receive an order confirmation within one working day. The order cannot be modified after receipt of the order confirmation.
Are you looking for specific products, alternatives or documentation?